Heat energy absorbed by a system in going through a cyclic process shown in figure is
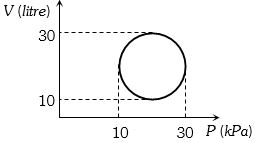

AIIMS 1995, Medium
(c) In a cyclic, $\Delta U = 0 $
From FLOT, $\Delta Q = \Delta U + \Delta W = 0 + \Delta W = $ Area of closed curve
==> $\Delta$ Q = $\pi r^2$ = $\pi {\left( {\frac{{20}}{2}} \right)^2}k{P_a} \times litre$ $ = 100\,\pi \times {10^3} \times {10^{ - 3}}J = 100\,\pi J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure
- 2Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 3One mole of an ideal gas at initial temperature $T$, undergoes a quasi-static process during which the volume $V$ is doubled. During the process, the internal energy $U$ obeys the equation $U=a V^3$, where $a$ is a constant. The work done during this process isView Solution
- 4An engine has an efficiency of $0.25$ when temperature of sink is reduced by $58\,^oC$, if its efficiency is doubled, then the temperature of the source is ..... $^oC$View Solution
- 5A litre of dry air at $STP$ expands adiabatically to a volume of $3$ litres. If $\gamma=1.40,$ the work done by air is$(3^{1.4}=4.6555)$ [Take air to be an ideal gas $]$View Solution
- 6View SolutionA cycle tyre bursts suddenly. This represents an
- 7View SolutionA container that suits the occurrence of an isothermal process should be made of
- 8View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 9A balloon filled with helium $\left(32^{\circ} C \right.$ and $1.7\; atm$.) bursts. Immediately afterwards the expansion of helium can be considered asView Solution
- 10View SolutionThe slopes of isothermal and adiabatic curves are related as
