A gas is enclosed in a vessel at a constant temperature at a pressure of $5$ atmosphere and volume $4$ litre. Due to a leakage in the vessel, after some time, the pressure is reduced to $4$ atmosphere. As a result, the
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionVelocity of sound measured at a given temperature in oxygen and hydrogen is in the ratio
- 2A closed hollow insulated cylinder is filled with gas at ${0^o}C$ and also contains an insulated piston of negligible weight and negligible thickness at the middle point. The gas on one side of the piston is heated to ${100^o}C.$ If the piston moves $5\,cm,$ the length of the hollow cylinder is ..... $cm$View Solution
- 3The internal energy of $10\,g$ of nitrogen at $S.T.P$, is about ......... $J$View Solution
- 4An ideal gas $(\gamma = 1.5)$ is expanded adiabatically. How many times has the gas to be expanded to reduce the root mean square velocity of molecules $2.0$ timesView Solution
- 5Two ideal gases at absolute temperature $T_1$ and $T_2$ are mixed. There is no loss of energy. The masses of the molecules are $m_1$ and $m_2$ and the number of molecules in the gases are $n_1$ and $n_2$ respectively. The temperature of mixture will beView Solution
- 6If $r.m.s.$ velocity of a gas is ${V_{rms}} = 1840m/s$ and its density $\rho = 8.99 \times {10^{ - 2}}kg/{m^3},$ the pressure of the gas will beView Solution
- 7Consider a gas for which diameter of molecules is $\sigma $ , the gas is at a pressure $P$ and temperature $T$ and $N_a =$ avagodro’s number. The mean free path along $x$ axis isView Solution
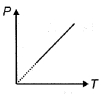
- 8The pressure $P$ of an ideal diatomic gas varies with its absolute temperature $T$ as shown in figure. The molar heat capacity of gas during this process is ........... $R$ [$R$ is gas constant]View Solution
- 9Two moles of ideal helium gas are in a rubber balloon at $30^o C$. The balloon is fully expandable and can be assumed to required no energy in its expansion. The temperature of the gas in the balloon is slowly changed to $35^o C$. The amount of heat required in raising the temperature is nearly...... $J$View Solution
(take $R$ $=$ $8.31$ $J/mol.K$) - 10A cylindrical container of volume $4.0 \times 10^{-3} \,{m}^{3}$ contains one mole of hydrogen and two moles of carbon dioxide. Assume the temperature of the mixture is $400 \,{K}$ The pressure of the mixture of gases is:View Solution
[Take gas constant as $8.3\, {J} {mol}^{-1} {K}^{-1}$]
