Consider a gas for which diameter of molecules is $\sigma $ , the gas is at a pressure $P$ and temperature $T$ and $N_a =$ avagodro’s number. The mean free path along $x$ axis is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$125\, ml$ of gas $A$ at $0.60$ atmosphere and $150\, ml$ of gas $B$ at $0.80$ atmosphere pressure at same temperature is filled in a vessel of $1$ litre volume. What will be the total pressure of mixture at the same temperature ............... $\mathrm{atmosphere}$View Solution
- 2Initially a gas of diatomic molecules is contained in a cylinder of volume $V _{1}$ at a pressure $P_{1}$ and temperature $250\, K$. Assuming that $25 \%$ of the molecules get dissociated causing a change in number of moles. The pressure of the resulting gas at temperature $2000\, K ,$ when contained in a volume $2 V _{1}$ is given by $P _{2}$. The ratio $\frac{P _{2}}{ P _{1}}$ is.View Solution
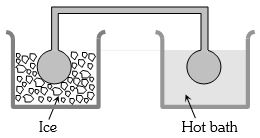
- 3Two identical glass bulbs are interconnected by a thin glass tube. A gas is filled in these bulbs at $N.T.P.$ If one bulb is placed in ice and another bulb is placed in hot bath, then the pressure of the gas becomes $1.5$ times. The temperature of hot bath will be ....... $^oC$View Solution
- 4From the following statements, concerning ideal gas at any given temperature $T,$ select the correct one(s)View Solution
- 5Following statements are given :View Solution
$(1)$ The average kinetic energy of a gas molecule decreases when the temperature is reduced.
$(2)$ The average kinetic energy of a gas molecule increases with increase in pressure at constant temperature.
$(3)$ The average kinetic energy of a gas molecule decreases with increases in volume.
$(4)$ Pressure of a gas increases with increase in temperature at constant pressure.
$(5)$ The volume of gas decreases with increase in temperature.
Choose the correct answer from the options given below:
- 6The average translational kinetic energy of ${N}_{2}$ gas molecules at $\ldots \ldots \ldots . .{ }^{\circ} {C}$ becomes equal to the ${K} . {E}$. of an electron accelerated from rest through a potential difference of $0.1$ $volt.$View Solution
$\left(\right.$ Given $\left.{k}_{{B}}=1.38 \times 10^{-23} \, {J} / {K}\right)$
(Fill the nearest integer).
- 7View SolutionSelect the incorrect statement about the specific heats of a gaseous system.
- 8View SolutionIn which of the following gas the root mean square velocity will be minimum (at const. temperature)
- 9A monoatomic gas of mass $4.0\, u$ is kept in an insulated container. Container is moving with velocity $30 \,m / s$. If container is suddenly stopped then change in temperature of the gas $\left( R =\right.$ gas constant) is $\frac{ x }{3 R } .$ Value of $x$ is ..........View Solution
- 10A box contains a mixture of ${H_2}$ and $He$ gases. Which of the following statements are correctsView Solution
