A gas is suddenly compressed to $1/4$ th of its original volume at normal temperature. The increase in its temperature is ....... $K$ $(\gamma = 1.5)$
Medium
(a) $\because \;T{V^{\gamma - 1}} = $constant $⇒$ ${T_1}V_1^{\gamma - 1} = {T_2}V_2^{\gamma - 1}$
$⇒$ ${T_2} = {T_1}{\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^{\gamma - 1}} = {T_1}{(4)^{1.5 - 1}} = 2{T_1}$
change in temperature
$ = {T_2} - {T_1} = 2{T_1} - {T_1} = {T_1} = 273\,K$
$⇒$ ${T_2} = {T_1}{\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^{\gamma - 1}} = {T_1}{(4)^{1.5 - 1}} = 2{T_1}$
change in temperature
$ = {T_2} - {T_1} = 2{T_1} - {T_1} = {T_1} = 273\,K$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas at atmospheric pressure is adiabatically compressed so that its density becomes $32$ times of its initial value. If the final pressure of gas is $128$ atmosphers, the value of $\gamma$ the gas isView Solution
- 2View SolutionFor a reversible process, necessary condition is
- 3A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 4A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
- 5Adiabatic modulus of elasticity of a gas is $2.1 \times {10^5}N/{m^2}.$ What will be its isothermal modulus of elasticity $\left( {\frac{{{C_p}}}{{{C_v}}} = 1.4} \right)$View Solution
- 6One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .View Solution
- 7A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
- 8View SolutionDuring an isothermal expansion of an ideal gas
- 9A cyclic process $ABCA$ is shown in the $V-T$ diagram. Process on the $P-V$ diagram isView Solution
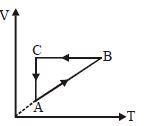
- 10An ideal gas is taken from point $A$ to the point $B,$ as shown in the $P-V$ diagram, keeping the temperature constant. The work done in the process isView Solution