An ideal gas at atmospheric pressure is adiabatically compressed so that its density becomes $32$ times of its initial value. If the final pressure of gas is $128$ atmosphers, the value of $\gamma$ the gas is
JEE MAIN 2013, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
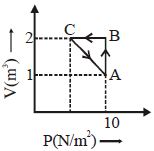
- 1A monoatomic gas is taken along path $AB$ as shown. Calculate change in internal energy of systemView Solution
- 2An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 3View SolutionThe adiabatic Bulk modulus of a diatomic gas at atmospheric pressure is
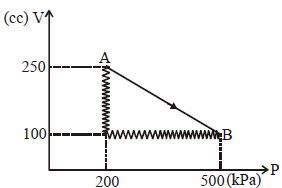
- 4The adjoining figure shows the $P-V$ diagram for a fixed mass of an ideal gas undergoing cyclic process. $AB$ represents isothermal process and $CA$ represents isochoric process.Which of the graph shown in the following figures represents the $P-T$ diagram of the cyclic process ?View Solution
- 5A heat engine is involved with exchange of heat of $1915\, J,$ $-40\, J ,+125\, J$ and $-Q\,J$, during one cycle achieving an efficiency of $50.0 \%$. The value of $Q$ is.......$J$View Solution
- 6View SolutionIn Carnot engine the work done by working substance is equivalent to
- 7An engine runs between a reservoir at temperature $200 \,K$ and a hot body which is initially at temperature of $600 \,K$. If the hot body cools down to a temperature of $400 \,K$ in the process, then the maximum amount of work that the engine can do (while working in a cycle) is (the heat capacity of the hot body is $1 \,J / K )$View Solution
- 8In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
- 9A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)View Solution
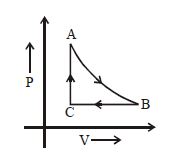
- 10An ideal gas is taken through the cycle $A \to B \to C \to A,$ as shown in the figure. What is the change in internal energy ...... $J$.View Solution