A gas mixture consists of $2$ moles of $O_2$ and $4$ moles of $Ar$ at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system is
IIT 1999,NEET 2017, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
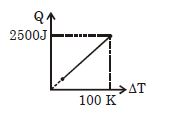
- 1One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
- 2Three perfect gases at absolute temperatures $T_1 , T_2$ and $T_3$ are mixed. The masses of molecules are $m_1 , m_2$ and $m_3$ and the number of molecules are $n_1, n_2$ and $n_3$ respectively. Assuming no loss of energy, the final temperature of the mixture isView Solution
- 3The temperature of an ideal gas is reduced from $927^\circ C$ to $27^\circ C$. The $r.m.s.$ velocity of the molecules becomesView Solution
- 4When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
- 5Two ideal polyatomic gases at temperatures $T _{1}$ and $T _{2}$ are mixed so that there is no loss of energy. If $F _{1}$ and $F _{2}, m _{1}$ and $m _{2}, n _{1}$ and $n _{2}$ be the degrees of freedom, masses, number of molecules of the first and second gas respectively, the temperature of mixture of these two gases isView Solution
- 6The ratio of two specific heats $\frac{{{C_P}}}{{{C_V}}}$ of $CO$ isView Solution
- 7One litre of oxygen at a pressure of $1\, atm$ and two litres of nitrogen at a pressure of $0.5\, atm$ are introduced into a vessel of volume $1L$. If there is no change in temperature, the final pressure of the mixture of gas (in $atm$) isView Solution
- 8An ideal gas of Molar mass $M$ is contained in a vertical tube of height $H$, closed at both ends. The tube is accelerating vertically upwards with acceleration $g$ Then, the ratio of pressure at the bottom and the mid point of the tube will beView Solution
- 9At $10^o C$ the value of the density of a fixed mass of an ideal gas divided by it pressure is $x.$ At $110^o C$ this ratio isView Solution
- 10A mixture of $2$ moles of helium gas (atomic mass $=4 \ amu$ ), and $1$ mole of argon gas (atomic mass $=40 \ amu$ ) is kept at $300 \ K$ in a container. The ratio of the rms speeds $\left(\frac{\left. v _{ mms } \text { (helium }\right)}{ v _{\text {rms }} \text { (argon) }}\right)$ is :View Solution
