A gas mixture consists of molecules of type $1, 2$ and $3$, with molar masses ${m_1} > {m_2} > {m_3}.$ ${V_{rms}}$ and $\overline K $ are the $r.m.s.$ speed and average kinetic energy of the gases. Which of the following is true
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At constant volume the specific heat of a gas is $\frac{{3R}}{2}$, then the value of $'\gamma '$ will be ....View Solution
- 2The average translational energy and the $r.m.s.$ speed of molecules in a sample of oxygen gas at $300 K$ are $6.21 \times {10^{ - 21}}\,J$ and $484\, m/s$ respectively. The corresponding values at $600 K$ are nearly (assuming ideal gas behaviour)View Solution
- 3On giving equal amount of heat at constant volume to $1\, mol$ of a monoatomic and a diatomic gas the rise in temperature $(\Delta T)$ is more forView Solution
- 4If the molecular weight of two gases are $M_1$ and $ M_1$, then at a temperature the ratio of root mean square velocity $v_1$ and $v_2$ will beView Solution
- 5A $15\,g$ mass of nitrogen gas is enclosed in a vessel at a temperature $27\,^oC.$ Amount of heat transferred to the gas, so that rms velocity of molecules is doubled, is about ...... $kJ$ [Take $R = 8.3\,J/K\,mole$ ]View Solution
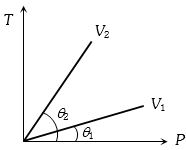
- 6From the following $P-T$ graph what interference can be drawnView Solution
- 7A flat plate is moving normal to its plane through a gas under the action of constant force $F$. The gas is kept at a very low pressure. The speed of the plate $v$ is much less than the average speed $u$ of the gas molecules. Which of the following options is/are true?View Solution
[$A$] The resistive force experienced by the plate is proportional to $\mathrm{v}$
[$B$] The pressure difference between the leading and trailing faces of the plate is proportional to uv
[$C$] The plate will continue to move with constant non-zero acceleration, at all times
[$D$] At a later time the external force $F$ balances the resistive force.
- 8When water is heated from $0$ to $4\,^oC$View Solution
- 9A mixture of $2$ moles of helium gas (atomic mass $=4 \ amu$ ), and $1$ mole of argon gas (atomic mass $=40 \ amu$ ) is kept at $300 \ K$ in a container. The ratio of the rms speeds $\left(\frac{\left. v _{ mms } \text { (helium }\right)}{ v _{\text {rms }} \text { (argon) }}\right)$ is :View Solution
- 10The graph between pressure $(P)$ and $\frac{1}{V}$ ($V$ is volume) for constant temperature process in ideal equation $(PV = nRT)$View Solution
