The average translational energy and the $r.m.s.$ speed of molecules in a sample of oxygen gas at $300 K$ are $6.21 \times {10^{ - 21}}\,J$ and $484\, m/s$ respectively. The corresponding values at $600 K$ are nearly (assuming ideal gas behaviour)
IIT 1997, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A container $X$ has volume double that of contianer $Y$ and both are connected by a thin tube. Both contains same ideal gas. The temperature of $X$ is $200\,\,K$ and that of $Y$ is $400\,\,K$. If mass of gas in $X$ is $m$ then in $Y$ it will be:View Solution
- 2View SolutionA diatomic molecule has how many degrees of freedom
- 3The pressure $P,$ volume $V$ and temperature $T$ of a gas in the jar $A$ and the other gas in the jar $B$ at pressure $2P,$ volume $V/4$ and temperature $2T,$ then the ratio of the number of molecules in the jar $A$ and $B$ will beView Solution
- 4The kinetic energy of one gram molecule of a gas at normal temperature and pressure is $(R = 8.31\,J/mole - K)$View Solution
- 5View SolutionWhich of the following statement is true
- 6A box contains n molecules of a gas. How will the pressure of the gas be effected, if the number of molecules is made $2n$View Solution
- 7The temperature of an ideal gas is increased from $200\,K$ to $800\,K$. If r.m.s. speed of gas at $200\,K$ is $v_0$. Then, r.m.s. speed of the gas at $800\,K$ will be:View Solution
- 8Figure shows two flasks connected to each other. The volume of the flask $1$ is twice that of flask $2.$ The system is filled with an ideal gas at temperature $100\, K$ and $200 \,K $ respectively. If the mass of the gas in $1$ be $m$ then what is the mass of the gas in flask $2$View Solution
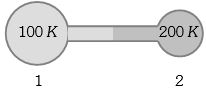
- 9Two vessels separately contain two ideal gases $A$ and $B$ at the same temperature, the pressure of $A$ being twice that of $B.$ Under such conditions, the density of $A$ is found to be $1.5$ times the density of $B.$ The ratio of molecular weight of $A$ and $B$ isView Solution
- 10The ratio of average translational kinetic energy to rotational kinetic energy of a diatomic molecule at temperature $T$ isView Solution
