When water is heated from $0$ to $4\,^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
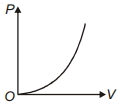
- 1The variation of pressure $P$ with volume $V$ for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process isView Solution
- 2$105$ calories of heat is required to raise the temperature of $3$ moles of an ideal gas at constant pressure from $30^{\circ} C$ to $35^{\circ} C$. The amount of heat required in calories to raise the temperature of the gas through the range $\left(60^{\circ} C\right.$ to $\left.65^{\circ} C \right)$ at constant volume is ........ $cal$ $\left(\gamma=\frac{C_p}{C_v}=1.4\right)$View Solution
- 3View SolutionThe degrees of freedom of a triatomic gas is
- 4When temperature of an ideal gas is increased from $27^\circ C$ to $227^\circ C$, its $r.m.s.$ speed changed from $400\, metre/sec$ to ${V_s}.$ The ${V_s}$ is ........ $metre/sec$View Solution
- 5View SolutionAccording to the kinetic theory of gases, at absolute temperature
- 6Two gases are at absolute temperatures $300 K $ and $350 K$ respectively. Ratio of average kinetic energy of their molecules isView Solution
- 7Five particles have speeds $1, 2, 3, 4, 5 \,\,m/s$. the average velocity of the particles is (in $m/s$)View Solution
- 8The temperature at which the root mean square velocity of a molecule will be doubled than at $100°C$View Solution
- 9We write the relation for Boyle's law in the form $PV = C$ when the temperature remains constant. In this relation, the magnitude of $C$ depends uponView Solution
- 10The value of $\gamma\left(=\frac{\mathrm{C}_{\mathrm{p}}}{\mathrm{C}_{\mathrm{v}}}\right),$ for hydrogen, hellium and another ideal diatomic gas $X$(whose molecules are not rigid but have an additional vibrational mode), are respectively equal toView Solution
