The kinetic energy of one gm-mole of a gas at normal temperature and pressure is $(R = 8.31 J/Mole-K)$
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If mass of $He$ atom is $4$ times that of hydrogen atom then mean velocity of $He$ isView Solution
- 2For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution
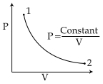
- 3At what temperature the molecules of nitrogen will have the same $r.m.s.$ velocity as the molecules of oxygen at $127°C$ ....... $^oC$View Solution
- 4View SolutionThe r.m.s. speed of gas molecules is given by
- 5When pressure remaining constant, at what temperature will the $r.m.s.$ speed of a gas molecules increase by $10 \%$ of the r.m.s. speed at $STP$?View Solution
- 6At what temperature will the $rms$ speed of oxygen molecules become just sufficient for escaping from the Earth's atmosphere $?$ (Given: Mass of oxygen molecule $(m)= 2.76 \times 10^{-26}\,kg$ Boltzmann's constant $k_B= 1.38 \times 10^{-23}\,\, JK^{-1}$)View Solution
- 7The kinetic energy of one gram molecule of a gas at normal temperature and pressure is $(R = 8.31\,J/mole - K)$View Solution
- 8The volume of a gas will be double of what it is at $0°C$ (pressure remaining constant) atView Solution
- 9The average kinetic energy of a monatomic molecule is $0.414 \mathrm{eV}$ at temperature :View Solution
(Use $\mathrm{K}_{\mathrm{B}}=1.38 \times 10^{-23} \mathrm{~J} / \mathrm{mol}-\mathrm{K}$ )
- 10The kinetic energy of translation of $20\, gm$ of oxygen at $47°C$ is (molecular wt. of oxygen is $32 \,gm/mol$ and $R = 8.3\, J/mol/K)$View Solution
