Internal forces acting in a system of particle can change
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$125\, ml$ of gas $A$ at $0.60$ atmosphere and $150\, ml$ of gas $B$ at $0.80$ atmosphere pressure at same temperature is filled in a vessel of $1$ litre volume. What will be the total pressure of mixture at the same temperature ............... $\mathrm{atmosphere}$View Solution
- 2The temperature at which the $r.m.s.$ speed of hydrogen molecules is equal to escape velocity on earth surface, will be ...... $K$View Solution
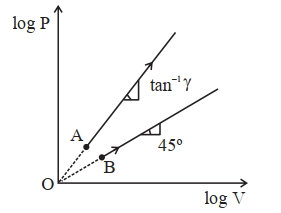
- 3Two thermodynamical process are shown in the figure. The molar heat capacity for process $A$ and $B$ are $C_A$ and $C_B$. The molar heat capacity at constant pressure and constant volume are represented by $C_P$ and $C_V$, respectively. Choose the correct statement.View Solution
- 4To what temperature should the hydrogen at $327°C$ be cooled at constant pressure, so that the root mean square velocity of its molecules become half of its previous value ....... $^oC$View Solution
- 5The molecules of a given mass of a gas have $r.m.s.$ velocity of $200 \,m s^{-1}$ at $27^o C$ and $1.0 \times 10^5 \,Nm^{-2}$ pressure. When the temperature and pressure of the gas are respectively, $127^o C$ and $0.05 \times 10^5 \,Nm^{-2},$ the $r.m.s.$ velocity of its molecules in $m s^{-1}$ isView Solution
- 6The temperature at which the root mean square velocity of a molecule will be doubled than at $100°C$View Solution
- 7View SolutionThe average kinetic energy of a gas molecule can be determined by knowing
- 8Vessel $A$ is filled with hydrogen while vessel $B,$ whose volume is twice that of $A,$ is filled with the same mass of oxygen at the same temperature. The ratio of the mean kinetic energies of hydrogen and oxygen isView Solution
- 9The root mean square speed of hydrogen molecules at $300$ $K$ is $1930\, m/s.$ Then the root mean square speed of oxygen molecules at $900\, K$ will be ....... $m/s$View Solution
- 10If radius of $O _{2}$ molecule $=40 \mathring A, T =27^{\circ} C$ and $P =1 \,atm .$ Find the time of relaxation.View Solution
