One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric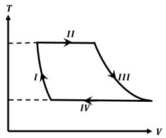
IIT 2018, Medium
$(A)$ Volume $\mathrm{V}$ is decreasing in process $\mathrm{I}$.
$(B)$ $\Delta \mathrm{U}=0, \mathrm{~W}>0$
$\Delta \mathrm{Q}>0$
Process II is isothermal expansion.
$(C)$ $\Delta \mathrm{U}=0, \mathrm{~W}<0$
$\Delta \mathrm{Q}<0$
Process $III$ is isothermal compression.
$(D)$ For an isobaric process $T-\mathrm{V}$ graph must be linear.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
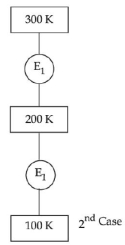
- 1In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution
- 2A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$View Solution
- 3A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
- 4Adiabatic modulus of elasticity of a gas is $2.1 \times {10^5}N/{m^2}.$ What will be its isothermal modulus of elasticity $\left( {\frac{{{C_p}}}{{{C_v}}} = 1.4} \right)$View Solution
- 5The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$View Solution
- 6View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter
- 7A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
- 8A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............View Solution
- 9View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
- 10If the ratio of specific heat of a gas at constant pressure to that at constant volume is $\gamma $, the change in internal energy of a mass of gas, when the volume changes from $V$ to $2V$ constant pressure $ p$, isView Solution
