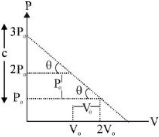
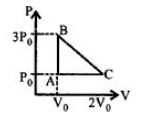
'$n$' moles of an ideal gas undergoes a process $A \rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will be

JEE MAIN 2016, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,View Solution
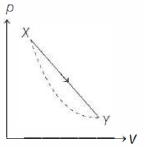
- 2In the $p-V$ diagram below, the dashed curved line is an adiabat.For a process that is described by a straight line joining two points $X$ and $Y$ on the adiabat (solid line in the diagram) heat is (Hint consider the variation in temperature from $X$ to $Y$ along the straight line)View Solution
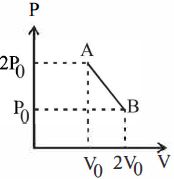
- 3One mole of an ideal monoatomic gas is taken along the path $ABCA$ as shown in the $PV$ diagram. The maximum temperature attained by the gas along the path $BC$ is given byView Solution
- 4View SolutionIrreversible process is
- 5View SolutionThe maximum possible efficiency of a heat engine is ...........
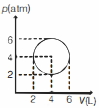
- 6An ideal gas undergoes a circular cycle centred at $4 \,atm , 4 L$ as shown in the diagram. The maximum temperature attained in this process is close toView Solution
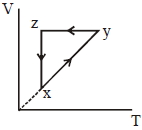
- 7A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
- 8A thermo-dynamical system is changed from state $({P_1},\,{V_1})$ to $({P_2},\,{V_2})$ by two different process. The quantity which will remain same will beView Solution
- 9A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
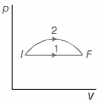
- 10An ideal gas undergoes change in its state from the initial state $I$ to the final state $F$ via two possible paths as shown below. Then,View Solution