A monoatomic gas is taken through a process $TP^{-1/3} =$ constant. If heat is given to the gas
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas with constant heat capacity $C_V=\frac{3}{2} n R$ is made to carry out a cycle that is depicted by a triangle in the figure given below. The following statement is true about the cycle.View Solution
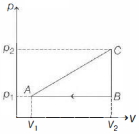
- 2View SolutionIn the above thermodynamic process, the correct statement is
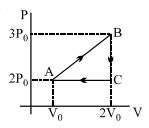
- 3An ideal gas is taken from point $A$ to the point $B,$ as shown in the $P-V$ diagram, keeping the temperature constant. The work done in the process isView Solution
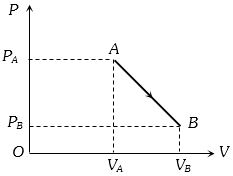
- 4$PV$ curve for the process whose $VT$ curve isView Solution
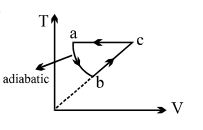
- 5An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution
- 6If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$View Solution
- 7$V\, = \,K\,{\left( {\frac{P}{T}} \right)^{0.33}}$ where $k$ is constant. It is an,View Solution
- 8View SolutionWhich of the following statements is correct for any thermodynamic system
- 9A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 10A bicycle tyre is filled with air having pressure of $270\,kPa$ at $27^{\circ}\,C$. The approximate pressure of the air in the tyre when the temperature increases to $36^{\circ}\,C$ is $............kPa$View Solution
