A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
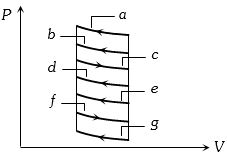
- 1The $P-V$ diagram shows seven curved paths (connected by vertical paths) that can be followed by a gas. Which two of them should be parts of a closed cycle if the net work done by the gas is to be at its maximum valueView Solution
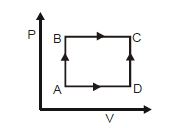
- 2A shown in the figure, the amount of heat absorbed along the path $ABC$ is $90\,J$ and the amount of work done by the system is $30\,J$ . If the amount of work done along the path $ADC$ is $20\,J$ , the amount of heat absorbed will be .... $J$View Solution
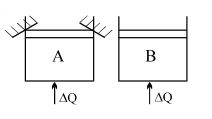
- 3Two identical vessels $A \& B$ contain equal amount of ideal monoatomic gas. The piston of $A$ is fixed but that of $B$ is free. Same amount of heat is absorbed by$A \& B$. If $B'$s internal energy increases by $100 \,\,J$ the change in internal energy of $A$ is ...... .$J$View Solution
- 4The efficiency of Carnot's engine operating between reservoirs, maintained at temperatures ${27^o}C$ and $ - {123^o}C,$ is ...... $\%$View Solution
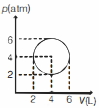
- 5An ideal gas undergoes a circular cycle centred at $4 \,atm , 4 L$ as shown in the diagram. The maximum temperature attained in this process is close toView Solution
- 6In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
- 7Three moles of an ideal gas $\left( {{C_P} = \frac{7}{2}R} \right)$ at pressure ${P_A}$ and temperature ${T_A}0$ is isothermally expanded to twice its initial volume. It is then compressed at constant pressure to its original volume. Finally the gas is compressed at constant volume to its original pressure ${P_A}.$ The correct $P-V$ and $P-T$ diagrams indicating the process areView Solution
- 8A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
- 9'$n$' moles of an ideal gas undergoes a process $A \rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will beView Solution
- 10A mixture of gases at $STP$ for which $\gamma=1.5$ is suddenly compressed to $\frac{1}{9}$ th of its original volume. The final temperature of mixture is .......... $^{\circ} C$View Solution
