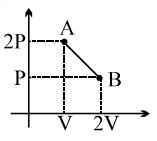
A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectively

Diffcult
$(\mathrm{AB}) \Rightarrow \mathrm{V}=\mathrm{constant}$
$\mathrm{W}_{\mathrm{AB}}=0$
$(\mathrm{BC}) \Rightarrow \mathrm{T}=\mathrm{constant}$
$\mathrm{W}=\mathrm{nRT} \ell n \left(\frac{\mathrm{V}_{2}}{\mathrm{V}_{1}}\right)$
$=\operatorname{RT} \ell n \left(\frac{V_{2}}{V_{1}}\right)$
$(\mathrm{CA}) \Rightarrow \mathrm{P}=\mathrm{constant}$
$\mathrm{W}=\mu \mathrm{R} \Delta \mathrm{T}$
$=\mathrm{P}_{1} \mathrm{V}_{1}-\mathrm{P}_{2} \mathrm{V}_{2}$
$=\mathrm{P}_{1}\left(\mathrm{V}_{1}-\mathrm{V}_{2}\right)$
$=\frac{\mathrm{RT}_{1}}{\mathrm{V}_{1}}\left(\mathrm{V}_{1}-\mathrm{V}_{2}\right)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
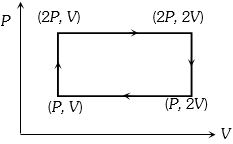
- 1Work done in the given $P-V$ diagram in the cyclic process isView Solution
- 2A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
- 3View SolutionThe internal energy of an ideal gas depends upon
- 4Heat energy of $735\,J$ is given to a diatomic gas allowing the gas to expand at constant pressure. Each gas molecule rotates around an internal axis but do not oscillate. The increase in the internal energy of the gas will be $..........\,J$View Solution
- 5View SolutionWhen an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is
- 6View SolutionFirst law thermodynamics states that
- 7Find the amount of work done to increase the temperature of one mole of an ideal gas by $30^o\ C$ if it is expanding under the condition .... $J$View Solution
$V\propto {T^{\frac{2}{3}}}$ $[R = 1.99\ cal/mol-K]$
- 8A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
- 9When $1\, gm$ of water at ${0^o}C$ and $1 \times {10^5}\;N/{m^2}$ pressure is converted into ice of volume $1.091\;c{m^2}$, the external work done will beView Solution
- 10One mole of a gas expands obeying the relation as shown in the $P/V$ diagram. The maximum temperature in this process is equal toView Solution