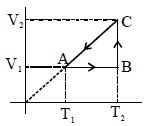
A Carnot engine with efficiency $50\,\%$ takes heat from a source at $600\,K$. In order to increase the efficiency to $70\,\%$, keeping the temperature of sink same, the new temperature of the source will be $.........\,K$
JEE MAIN 2023, Medium
$\text { Initially } \eta=\frac{1}{2}$
$\eta=1-\frac{ T _2}{ T _1}$
$\therefore \frac{1}{2}=1-\frac{ T _2}{600} \quad \Rightarrow T _2=300\,K$
$\Rightarrow \frac{ T _2}{600}=\frac{1}{2} \quad$
Now efficiency is increased to $70 \%$ and $T _2=300$ $K$, Let temp of source $T _1= T$
$\Rightarrow \frac{7}{10}=1-\frac{300}{T}$
$\Rightarrow \frac{300}{ T }=1-\frac{7}{10}$
$\Rightarrow \frac{300}{ T }=\frac{3}{10} \quad \therefore T =1000\,K$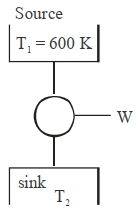
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1During an experiment, an ideal gas is found to obey a condition $\frac{{{P^2}}}{\rho }$ = constant [$\rho =$ density of the gas]. The gas is initially at temperature $T,$ pressure $P$ and density $\rho$ . The gas expands such that density changes to $\rho/2$View Solution
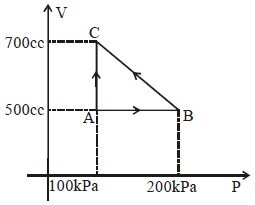
- 2A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
- 3The temperature inside and outside of refrigerator are $260\, K$ and $315\, K$ respectively. Assuming that the refrigerator cycle is reversible, calculate the heat delivered to surroundings for every joule of work done.View Solution
- 4Heat energy of $735\,J$ is given to a diatomic gas allowing the gas to expand at constant pressure. Each gas molecule rotates around an internal axis but do not oscillate. The increase in the internal energy of the gas will be $..........\,J$View Solution
- 5View SolutionNeon gas of a given mass expands isothermally to double volume. What should be the further fractional decrease in pressure, so that the gas when adiabatically compressed from that state, reaches the original state?
- 6Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$View Solution
- 7An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
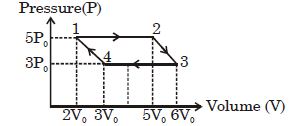
- 8A sample of gas at temperature $\mathrm{T}$ is adiabatically expanded to double its volume. Adiabatic constant for the gas is $\gamma=3 / 2$. The work done by the gas in the process is : $(\mu=1 \mathrm{~mole})$View Solution
- 9View SolutionThe internal energy of an ideal gas depends upon
- 10A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectivelyView Solution