A partition divides a container having insulated walls into two compartments $I$ and $II$. the same gas fills the two compartments. The ratio of the number of molecules in compartments $I$ and $II$ is


Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe ratio of mean kinetic energy of hydrogen and oxygen at a given temperature is
- 2Two non-reactive monoatomic ideal gases have their atomic masses in the ratio $2: 3$. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is $4: 3$. The ratio of their densities is:View Solution
- 3$\frac{1}{2} $ mole of helium gas is contained in a container at $S.T.P.$ The heat energy needed to double the pressure of the gas, keeping the volume constant (specific heat of the gas $ = 3\,J\,g{m^{ - 1}}\,{K^{ - 1}})$ is ...... $J$View Solution
- 4The value closest to the thermal velocity of a Helium atom at room temperature $(300\,K)$in $ms^{-1}$ is $[k_B\, = 1 .4\times10^{-23}\,J/K;\, m_{He}\, = 7\times10^{-27}\,kg]$View Solution
- 5Nitrogen gas is at $300^{\circ} C$ temperature. The temperature (in $K$) at which the $rms$ speed of a $H _{2}$, molecule would be equal to the $rms$ speed of a nitrogen molecule, is........View Solution
(Molar mass of $N _{2}$ gas $28\, g$ )
- 6Pressure $Vs$ temperature graph of an ideal gas is shown in figure. Density of the gas at point $A$ is $\rho _0$ , Density at $B$ will beView Solution
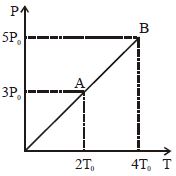
- 7A constant pressure air thermometer gave a reading of $47.5$ units of volume when immersed in ice cold water, and $67$ units in a boiling liquids. The boiling point of the liquid will be .......... $^oC$View Solution
- 8View SolutionAccording to the kinetic theory of gases, total energy of a gas is equal to
- 9Considering the gases to be ideal, the value of $\gamma = \frac{{{C_P}}}{{{C_V}}}$ for a gaseous mixture consisting of $= 3$ moles of carbon dioxide and $2$ moles of oxygen will be $({\gamma _{{O_2}}} = 1.4,\,\,{\gamma _{C{O_2}}} = 1.3)$View Solution
- 10An ideal gas in a closed container is slowly heated. As its temperature increases, which of the following statements are true$?$View Solution
$(A)$ the mean free path of the molecules decreases.
$(B)$ the mean collision time between the molecules decreases.
$(C)$ the mean free path remains unchanged.
$(D)$ the mean collision time remains unchanged.
