Nitrogen gas is at $300^{\circ} C$ temperature. The temperature (in $K$) at which the $rms$ speed of a $H _{2}$, molecule would be equal to the $rms$ speed of a nitrogen molecule, is........
(Molar mass of $N _{2}$ gas $28\, g$ )
JEE MAIN 2020, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionAn ideal gas is filled in a vessel, then
- 2By what factor the $r.m.s.$ velocity will change, if the temperature is raised from $27^\circ C$ to $327^\circ C$View Solution
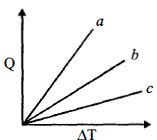
- 3Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond toView Solution
- 4A gas is filled in a cylinder, its temperature is increased by $20\%$ on Kelvin scale and volume is reduced by $10\%.$ How much percentage of the gas will leak out ........ $\%$View Solution
- 5A soap bubble of radius $r$ has monoatomic ideal- gas inside. The gas is heated in such a manner that bubble remains in mechanical equillibrium. Assuming that the soap material of the bubble has no heat capacity, the molar heat capacity of the gas in the process will beView Solution
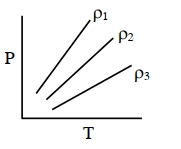
(Neglect atmospheric pressure) - 6$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :View Solution
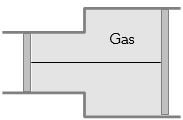
- 7View SolutionA gas is filled in the cylinder shown in the figure. The two pistons are joined by a string. If the gas is heated, the pistons will
- 8Hydrogen gas is filled in a balloon at $20^\circ C$. If temperature is made $40^\circ C$, pressure remaining same, what fraction of Hydrogen will come outView Solution
- 9The average distance between molecules of an ideal gas at $STP$ is approximately of the order ofView Solution
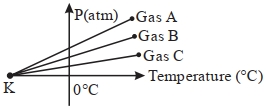
- 10For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.View Solution
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$