A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$
NEET 2016, Medium
$Given,\,{T_2} = {4^ \circ }C = 277\,K,{T_1} = {30^ \circ }C = 303\,K$
${Q_2} = 600\,cal\,per\,second$
Coefficient of performance, $\alpha = \frac{{{T_2}}}{{{T_1} - {T_2}}}$
$ = \frac{{277}}{{303 - 277}} = \frac{{277}}{{26}}$
Also,$\alpha = \frac{{{Q_2}}}{W}$
$\therefore $ $Work\,to\,be\,done\,per\,second=power\,required$
$ = W = \frac{{{Q_2}}}{\alpha } = \frac{{26}}{{277}} \times 600\,cal\,per\,second$
$ = \frac{{26}}{{277}} \times 600 \times 4.2\,J\,per\,second = 236.5\,W$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The volume of a gas is reduced adiabatically to $\frac{1}{4}$ of its volume at $27°C$, if the value of $\gamma = 1.4,$ then the new temperature will beView Solution
- 2View SolutionThe first operation involved in a Carnot cycle is
- 3Water falls from a height of $60\,m$ at the rate of $15\,kg/s$ to operate a turbine. The losses due to frictional forces are $10\%$ of energy. How much power is generated by the turbine ....... $kW$ $(g = 10\,m/s^2)$View Solution
- 4View SolutionIn the following figures heat is absorbed by the gas
- 5In the reported figure, heat energy absorbed by a system in going through a cyclic process is $......\,\pi J$View Solution
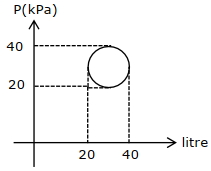
- 6A polyatomic gas with six degrees of freedom does $25\,\,J$ of work when it is expanded at constant pressure. The heat given to the gas is ..... $J$View Solution
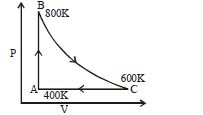
- 7One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 8An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution
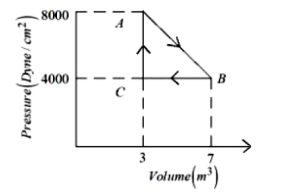
- 9A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :View Solution
- 10For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
