A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$
NEET 2016, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe first law of thermodynamics is concerned with the conservation of
- 2An ideal gas undergoes a four step cycle as shown in the $P-V$ diagram below. During this cycle, in which step heat is absorbed by the gasView Solution
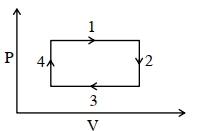
- 3If ${C_V} = 4.96cal/mole\, K$, then increase in internalenergy when temperature of $2$ moles of this gas is increased from $340 K$ to $342 K$ ....... $cal$View Solution
- 4$P-V$ diagram of an ideal gas is as shown in figure. Work done by the gas in process $ABCD$ isView Solution
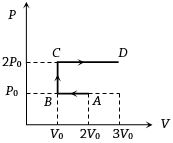
- 5Agas expands such that its initial and final temperature are equal. Also, the process followed by the gas traces a straight line on the $P-V$ diagram :View Solution
- 6The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
- 7In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 8A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
- 9When the amount of work done is $333\, cal$ and change in internal energy is $167\, cal$, then the heat supplied is ....... $cal$View Solution
- 10View SolutionIn a reversible isochoric change
