A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion is
AIIMS 1998,AIPMT 1997, Medium
(a) In thermodynamic process, work done is equal to the area covered by the $PV$ curve with volume axis.
Hence, according to graph shown
${W_{adiabatic}} < {W_{isothermal}} < {W_{isobaric}}$
Hence, according to graph shown
${W_{adiabatic}} < {W_{isothermal}} < {W_{isobaric}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$View Solution
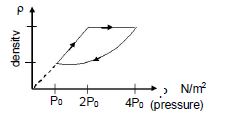
- 2An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
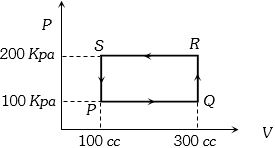
- 3A thermodynamic system is taken through the cycle $PQRSP$ process. The net work done by the system is ....... $J$View Solution
- 4In an Isobaric process, the work done by a di$-$atomic gas is $10\, J ,$ the heat given to the gas will be (in $J$)View Solution
- 5An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
- 6One mole of a gas obeying the equation of state $P(V-b)=R T$ is made to expand from a state with coordinates $\left(P_{1}, V_{1}\right)$ to a state with $\left(P_{2}, V_{2}\right)$ along a process that is depicted by a straight line on a $P-V$ diagram. Then, the work done is given byView Solution
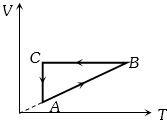
- 7A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
- 8View SolutionA gas undergoes a change at constant temperature. Which of the following quantities remain fixed?
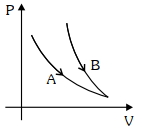
- 9Choose the correct statement for processes $A$ & $B$ shown in figure.View Solution
- 10When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
