One mole of a gas obeying the equation of state $P(V-b)=R T$ is made to expand from a state with coordinates $\left(P_{1}, V_{1}\right)$ to a state with $\left(P_{2}, V_{2}\right)$ along a process that is depicted by a straight line on a $P-V$ diagram. Then, the work done is given by
NEET 2017, Easy
Workdone during the complete cycle is equal to the area enclosed by the P-V graph
$W=\frac{1}{2} \text { base } \times \text { height }+ \text { Area of rectangular }$
$=\frac{1}{2}\left(V_2-V_1\right) \times\left(P_1-P_2\right)+\left(V_2-V_1\right) P_2$
$=\left(V_2-V_1\right)\left[\frac{P_1}{2}-\frac{P_2}{2}+P_2\right]$
$=\left(V_2-V_1\right)\left[\frac{P_1}{2}+\frac{P_2}{2}\right]$
$=\frac{1}{2}\left(P_1+P_2\right)\left(V_2-V_1\right)$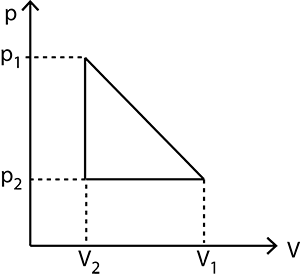
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
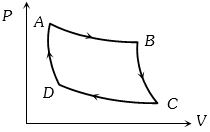
- 1The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
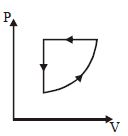
- 2For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
- 3The temperature of sink of Carnot engine is ${27^o}C$. Efficiency of engine is $25\%.$ Then temperature of source is ...... $^oC$View Solution
- 4One mole of an ideal gas expands adiabatically from an initial state $\left(T_A, V_0\right)$ to final state $\left(T_f, 5 V_0\right)$. Another mole of the same gas expands isothermally from a different initial state ( $T_{\mathrm{B}}, \mathrm{V}_0$ ) to the same final state $\left(T_{\mathrm{f}}, 5 V_0\right)$. The ratio of the specific heats at constant pressure and constant volume of this ideal gas is $\gamma$. What is the ratio $T_{\mathrm{A}} / T_{\mathrm{B}}$ ?View Solution
- 5View SolutionIn thermodynamics, heat and work are
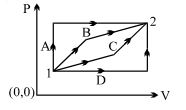
- 6An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
- 7An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will beView Solution
- 8A heat engine operates with the cold reservoir at temperature $324 K$. The minimum temperature of the hot reservoir, if the heat engine takes $300 \; J$ heat from the hot reservoir and delivers $180 \; J$ heat to the cold reservoir per cycle, is $\dots \; K .$View Solution
- 9A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
- 10Heat given to a system is $35$ joules and work done by the system is $15$ joules. The change in the internal energy of the system will be ..... $J$View Solution
