$\frac{W}{Q}=\frac{n R \Delta T}{n C_{p} \Delta T}$
$=\frac{n R \Delta T}{n\left(\frac{f}{2}+1\right) R \Delta T}$
$=\frac{1}{\frac{f}{2}+1}$ $........(I)$
Substitute $5$ for $f$ in equation $(I).$
$\frac{W}{Q}=\frac{1}{\frac{5}{2}+1}$
$=\frac{2}{7}$
$Q=\frac{7}{2} W$ $...(II)$
Substitute $10$ for $W$ in equation $(II).$
$Q=\frac{7}{2}(10)$
$=35 J$
Download our appand get started for free
Similar Questions
- 1View SolutionThe rate of recombination or generation are governed by the law(s) of
- 2The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
- 3An engine has an efficiency of $0.25$ when temperature of sink is reduced by $58\,^oC$, if its efficiency is doubled, then the temperature of the source is ..... $^oC$View Solution
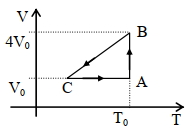
- 4One mole of an ideal gas in initial state $\mathrm{A}$ undergoes a cyclic process $A B C A$, as shown in the figure. Its pressure at $A$ is $\mathrm{P}_0$. Choose the correct option$(s)$ from the followingView Solution
$(A)$ Internal energies at $\mathrm{A}$ and $\mathrm{B}$ are the same
$(B)$ Work done by the gas in process $\mathrm{AB}$ is $\mathrm{P}_0 \mathrm{~V}_0 \ln 4$
$(C)$ Pressure at $C$ is $\frac{P_0}{4}$
$(D)$ Temperature at $\mathrm{C}$ is $\frac{\mathrm{T}_0}{4}$
- 5A Carnot engine with sink's temperature at $17\,^oC$ has $50\%$ efficiency. By how much should its source temperature be changed to increases its efficiency to $60\%$ ?...... $K$View Solution
- 6If $\Delta U$ and $\Delta W$ represent the increase in internal energy and work done by the system respectively in a thermodynamical process, which of the following is true?View Solution
- 7Given below are two statementView Solution
Statement $-I$ : What $\mu$ amount of an ideal gas undergoes adiabatic change from state $\left( P _{1}, V _{1}, T _{1}\right)$ to state $\left( P _{2}, V _{2}, T _{2}\right)$, the work done is $W =\frac{1 R \left( T _{2}- T _{1}\right)}{1-\gamma}$, where $\gamma=\frac{ C _{ P }}{ C _{ V }}$ and $R =$ universal gas constant,
Statement $-II$ : In the above case. when work is done on the gas. the temperature of the gas would rise.
Choose the correct answer from the options given below
- 8Heat is given to an ideal gas in an isothermal process.View Solution
$A.$ Internal energy of the gas will decrease.
$B.$ Internal energy of the gas will increase.
$C.$ Internal energy of the gas will not change.
$D.$ The gas will do positive work.
$E.$ The gas will do negative work.
Choose the correct answer from the options given below :
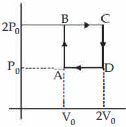
- 9Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)View Solution
- 10The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution