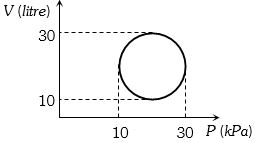
A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, then
AIPMT 2001, Medium
(a) ${\eta _{\max }} = 1 - \frac{{{T_2}}}{{{T_1}}} = 1 - \frac{{300}}{{400}} = \frac{1}{4} = 25\% $
So $26\%$ efficiency is impossible
So $26\%$ efficiency is impossible
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 2View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter
- 3The volume of $1\; mole$ of an ideal gas with the adiabatic exponent $\gamma$ is changed according to the relation $V=\frac bT$ where $b =$ constant. The amount of heat absorbed by the gas in the process if the temperature is increased by $\triangle T$ will beView Solution
- 4An ideal gas at a pressures of $1$ atmosphere and temperature of ${27^o}C$ is compressed adiabatically until its pressure becomes $8$ times the initial pressure, then the final temperature is ..... $^oC$ ($\gamma = 3/2$)View Solution
- 5A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 6An ideal gas goes from state $A$ to state $B$ via three different processes as indicated in the $P-V$ diagram. If $Q_1, Q_2, Q_3$ indicate the heat absorbed by the gas along the three processes and $\Delta U_1, \Delta U_2, \Delta U_3$View Solution
indicate the change in internal energy along the three processes respectively, then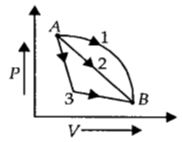
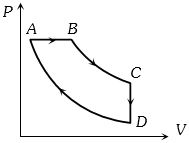
- 7A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
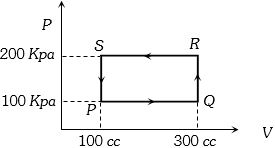
- 8A thermodynamic system is taken through the cycle $PQRSP$ process. The net work done by the system is ....... $J$View Solution
- 9A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process ..... $J$. ($J = 4.18$ joules/cal)View Solution
- 10View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is