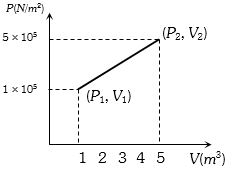
A system changes from the state $({P_1},\;{V_1})$ to $({P_2},{V_2})$ as shown in the figure. What is the work done by the system

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
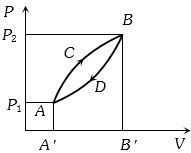
- 1A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the areaView Solution
- 2In an Isothermal change, the change in pressure and volume of a gas can be represented for three different temperature; $T _3 > T _2 > T _1$ as :View Solution
- 3A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphereView Solution
- 4A monoatomic ideal gas, initially at temperature ${T_1},$ is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature. ${T_2}$ by releasing the piston suddenly. If ${L_1}$ and ${L_2}$ are the lengths of the gas column before and after expansion respectively, then ${T_1}/{T_2}$ is given byView Solution
- 5In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $30$ joules of heat and $10$ joules of work was done on the gas. If the initial internal energy of the gas was $30$ joules, then the final internal energy will be ........ $J$View Solution
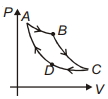
- 6Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution
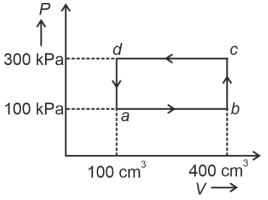
- 7A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ isView Solution
- 8This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1 :$ An inventor claims to have constructed an engine that has an efficiency of $30\%$ when operated between the boiling and freezing points of water. This is not possible.
Statement $2:$ The efficiency of a real engine is always less than the efficiency of a Carnot engine operating between the same two temperatures.
- 9The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution
- 10A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
