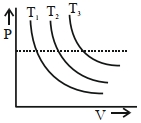
In an Isothermal change, the change in pressure and volume of a gas can be represented for three different temperature; $T _3 > T _2 > T _1$ as :
JEE MAIN 2023, Medium
For isothermal process $P - V$ graph is rectangular hyperbola As dotted line is isobaric line which implies $T _3 > T _2 > T _1$ as volume is increasing.

Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn thermodynamics, heat and work are
- 2Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
- 3An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
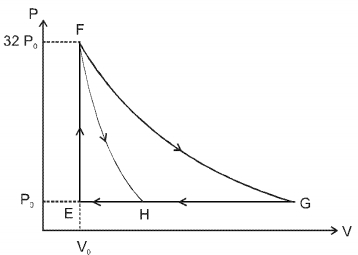
- 4One mole of a monatomic ideal gas is taken along two cyclic processes $E \rightarrow F \rightarrow G \rightarrow E$ and $E \rightarrow F \rightarrow H \rightarrow$ E as shown in the $PV$ diagram. The processes involved are purely isochoric, isobaric, isothermal or adiabatic. $Image$View Solution
Match the paths in List $I$ with the magnitudes of the work done in List $II$ and select the correct answer using the codes given below the lists.
List $I$ List $I$ $P.$ $\quad G \rightarrow E$ $1.$ $\quad 160 P_0 V_0 \ln 2$ $Q.$ $\quad G \rightarrow H$ $2.$ $\quad 36 P _0 V _0$ $R.$ $\quad F \rightarrow H$ $3.$ $\quad 24 P _0 V _0$ $S.$ $\quad F \rightarrow G$ $4.$ $\quad 31 P_0 V_0$ Codes: $ \quad \quad P \quad Q \quad R \quad S $
- 5Adiabatic modulus of elasticity of a gas is $2.1 \times {10^5}N/{m^2}.$ What will be its isothermal modulus of elasticity $\left( {\frac{{{C_p}}}{{{C_v}}} = 1.4} \right)$View Solution
- 6View SolutionA Camot cycle consists of
- 7For adiabatic processes $\left( {\gamma = \frac{{{C_p}}}{{{C_v}}}} \right)$View Solution
- 8A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution
- 9A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
- 10A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
