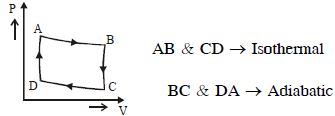Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal monoatomic gas expands to twice its volume. If the process is isothermal, the magnitude of work done by the gas is $W_i$. If the process is adiabatic, the magnitude of work done by the gas is $W_a$. Which of the following is true?View Solution
- 2In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
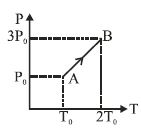
- 3Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
- 4View SolutionIn the following indicator diagram, the net amount of work done will be
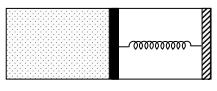
- 5An ideal monoatomic gas is confined in a horizontal cylinder by a spring loaded piston (as shown in the figure). Initially the gas is at temperature $T _1$, pressure $P_1$ and volume $V_1$ and the spring is in its relaxed state. The gas is then heated very slowly to temperature $T_2$, pressure $P _2$ and volume $V _2$. During this process the piston moves out by a distance $x$. Ignoring the friction between the piston and the cylinder, the correct statement$(s)$ is(are)View Solution
$(A)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the energy stored in the spring is $\frac{1}{4} P_1 V_1$
$(B)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the change in internal energy is $3 P_1 V_1$
$(C)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the work done by the gas is $\frac{7}{3} P_1 V_1$
$(D)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the heat supplied to the gas is $\frac{17}{6} P_1 V_1$
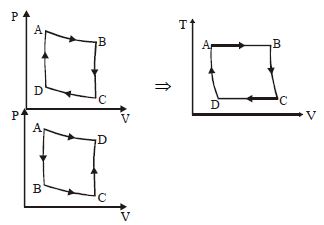
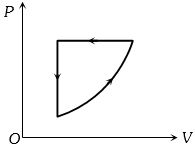
- 6For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correctView Solution
- 7A cylinder with a movable piston contains $3\,moles$ of hydrogen at standard temperature and pressure. The walls of the cylinder are made of a heat insulator, and the piston is insulated by having a pile of sand on it. By what factor does the pressure of the gas increases if the gas is compressed to half its original volume?View Solution
- 8A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 9A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
- 10If $\gamma = 2.5$ and volume is equal to $\frac{1}{8}$ times to the initial volume then pressure $P' $ is equal to (Initial pressure $= P$)View Solution