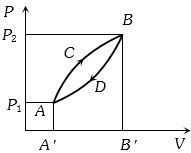
A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the area

AIPMT 1992, Medium
(c)The work done in cyclic process is equal to the area enclosed by the $PV$ diagram
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Starting with the same initial conditions, an ideal gas expands from volume $V_{1}$ to $V_{2}$ in three different ways. The work done by the gas is $W_{1}$ if the process is purely isothermal. $W _{2}$. if the process is purely adiabatic and $W _{3}$ if the process is purely isobaric. Then, choose the coned optionView Solution
- 2During an adiabatic compression, $830\, J$ of work is done on $2\, moles$ of a diatomic ideal gas to reduce its volume by $50\%$. The change in its temperahture is nearly..... $K$ $(R\, = 8.3\, J\,K^{-1}\, mol^{-1} )$View Solution
- 3View SolutionTwo gases are said to be in thermal equilibrium when they have same
- 4View SolutionThe internal energy of an ideal gas depends upon
- 5A gas expands under constant pressure $P$ from volume ${V_1}$ to ${V_2}$. The work done by the gas isView Solution
- 6A refrigerator consumes an average $35\, {W}$ power to operate between temperature $-10^{\circ} {C}$ to $25^{\circ} {C}$. If there is no loss of energy then how much average heat per second does it transfer? (in ${J} / {s}$)View Solution
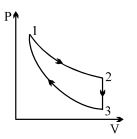
- 7Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
- 8The coefficient of performance of a refrigerator is $5.$ If the temperature inside freezer is $-20^o C,$ the temperature of the surroundings to which it rejects heat is ........ $^oC$View Solution
- 9View SolutionWhich of the following can not determine the state of a thermodynamic system
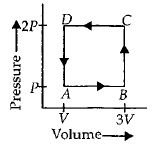
- 10A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution