A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $\gamma$. It is moving with speed $v$ and it's suddenly brought to rest. Assuming no heat is lost to the surroundings, its temperature increases by
AIEEE 2011, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Under constant temperature, graph between $P$ and $1/V$ isView Solution
- 2View SolutionMaxwell distribution function is shown in figure for different gases, which of the following is correct matching?
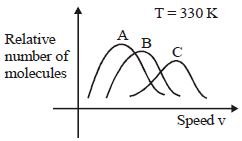
- 3View SolutionVariation of atmospheric pressure, with height from earth is ................
- 4The number of air molecules per $\mathrm{cm}^3$ increased from $3 \times 10^{19}$ to $12 \times 10^{19}$. The ratio of collision frequency of air molecules before and after the increase in number respectively is $.........$View Solution
- 5The pressure and volume of saturated water vapour are $P$ and $V$ respectively. It is compressed isothermally thereby volume becomes $V/2,$ the final pressure will beView Solution
- 6View SolutionA piston is slowly pushed into a metal cylinder containing an ideal gas. Which of the following statements is/are incorrect?
- 7A vessel of volume $8\, litre$ contains an ideal gas at $300\, K$ and $2\, atm$ pressure. The gas is allowed to leak till pressure become $125\, kpa$ calculate amount of moles which leak out if temperature remain constant ...... $moles$View Solution
- 8Under an adiabatic process, the volume of an ideal gas gets doubled. Consequently the mean collision time between the gas molecule changes from $\tau_{1}$ to $\tau_{2} .$ If $\frac{C_{p}}{C_{v}}=\gamma$ for this gas then a good estimate for $\frac{\tau_{2}}{\tau_{1}}$ is given by :View Solution
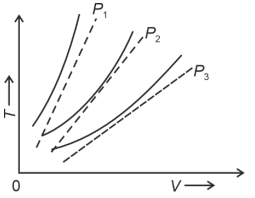
- 9The following graph represents the $T-V$ curves of an ideal gas (where $T$ is the temperature and $V$ the volume) at three pressures $P_1, P_2$ and $P_3$ compared with those of Charles's law represented as dotted lines.View Solution
Then the correct relation is:
- 10Two moles of ideal helium gas are in a rubber balloon at $30^o C$. The balloon is fully expandable and can be assumed to required no energy in its expansion. The temperature of the gas in the balloon is slowly changed to $35^o C$. The amount of heat required in raising the temperature is nearly...... $J$View Solution
(take $R$ $=$ $8.31$ $J/mol.K$)
