The following graph represents the $T-V$ curves of an ideal gas (where $T$ is the temperature and $V$ the volume) at three pressures $P_1, P_2$ and $P_3$ compared with those of Charles's law represented as dotted lines.
Then the correct relation is: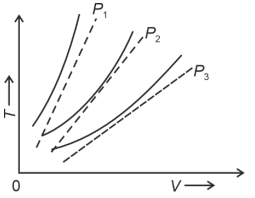
NEET 2024, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas $(\gamma = 1.5)$ is expanded adiabatically. How many times has the gas to be expanded to reduce the root mean square velocity of molecules $2.0$ timesView Solution
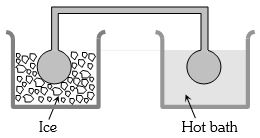
- 2Two identical glass bulbs are interconnected by a thin glass tube. A gas is filled in these bulbs at $N.T.P.$ If one bulb is placed in ice and another bulb is placed in hot bath, then the pressure of the gas becomes $1.5$ times. The temperature of hot bath will be ....... $^oC$View Solution
- 3The temperature at which the root mean square velocity of a molecule will be doubled than at $100°C$View Solution
- 4A gas at $27^o C$ has a volume $V$ and pressure $P.$ On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ...... $^oC$View Solution
- 5View SolutionSelect the incorrect statement about Maxwell's speed distribution
- 6$7$ mole of certain monoatomic ideal gas undergoes a temperature increase of $40 K$ at constant pressure. The increase in the internal energy of the gas in this process is$....J$ (Given $R =8.3 JK ^{-1} mol ^{-1}$ )View Solution
- 7At $10^o C$ the value of the density of a fixed mass of an ideal gas divided by it pressure is $x.$ At $110^o C$ this ratio isView Solution
- 8View SolutionOn colliding in a closed container the gas molecules
- 9In a vessel, the gas is at pressure $P_0$. If the mass of all the molecules is halved and their speed is doubled, then the resultant pressure will beView Solution
- 10At $100 \,K$ and $0.1$ atmospheric pressure, the volume of helium gas is $10 \,litres.$ If volume and pressure are doubled, its temperature will change to ....... $K$View Solution
