Under constant temperature, graph between $P$ and $1/V$ is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider a mixture of gas molecule of types $A, B$ and $C$ having masses $m_{A}\,<\,m_{B}\,<\,m_{C}$ ratio of their root mean square speeds at normal temperature and pressure is :View Solution
- 2An ideal gas $(\gamma = 1.5)$ is expanded adiabatically. How many times has the gas to be expanded to reduce the root mean square velocity of molecules $2.0$ timesView Solution
- 3Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ If the internal energy of He sample of $100\,\,J$ and that of the hydrogen sample is $200\,\,J$, then the internal energy of the mixture is ..... $J$View Solution
- 4In a thermally isolated system, two boxes filled with an ideal gas are connected by a valve. When the valve is in closed position, states of the box $1$ and $2$ respectively, are ( $1 \,atm , V, T)$ and $(0.5 \,atm , 4 V, T)$. When the valve is opened, then the final pressure of the system is approximately ............... $atm$View Solution
- 5To what temperature should the hydrogen at room temperature $(27°C)$ be heated at constant pressure so that the $R.M.S.$ velocity of its molecules becomes double of its previous value ....... $^oC$View Solution
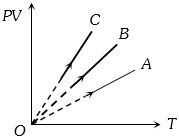
- 6$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
- 7A soap bubble of radius $r$ has monoatomic ideal- gas inside. The gas is heated in such a manner that bubble remains in mechanical equillibrium. Assuming that the soap material of the bubble has no heat capacity, the molar heat capacity of the gas in the process will beView Solution
(Neglect atmospheric pressure) - 8At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution
- 9The translational degrees of freedom $(f)$ and rotational degrees of freedom $(f)$ of $\mathrm{CH}_4$ molecule are:View Solution
- 10Consider a sample of oxygen behaving like an ideal gas. At $300 \,K ,$ the ratio of root mean square (rms) velocity to the average velocity of gas molecule would be :View Solution
(Molecular weight of oxygen is $32 \,g / mol$ $\left. R =8.3 \,J K ^{-1} mol ^{-1}\right)$
