($1$) The value of $\frac{T_R}{T_0}$ is
$(A)$ $\sqrt{2}$ $(B)$ $\sqrt{3}$ $(C)$ $2$ $(D)$ $3$
($2$) The value of $\frac{Q}{R T_0}$ is
$(A)$ $4(2 \sqrt{2}+1)$ $(B)$ $4(2 \sqrt{2}-1)$ $(C)$ $(5 \sqrt{2}+1)$ $(D)$ $(5 \sqrt{2}-1)$
Give the answer or qution ($1$) and ($2$)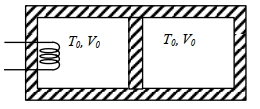
$C _{ V }=\frac{ R }{\gamma-1}=2 R \Rightarrow \gamma-1=\frac{1}{2}$
$\gamma=\frac{3}{2}$
$T _0 V _0^{\gamma-1}= T _{ R }\left(\frac{ V _0}{2}\right)^{\gamma-1}$
$\frac{ T _{ R }}{ T _0}=\sqrt{2}$
$\rho\left(\frac{ V _0}{2}\right)^\gamma= P _0 V _0^\gamma \Rightarrow P = P _0 \times 2^{\frac{3}{2}}$
$\frac{ PV }{ T _{ L }}=\frac{ P _0 V _0}{ T _0} \Rightarrow T _{ L }=2^{\frac{3}{2}} \times \frac{3}{2} T _0=3 \sqrt{2} T _0$
$Q = nC C _{ V } \Delta T _1+ nC V _{ V } \Delta T _2$
$=1 \times 2 R \times(3 \sqrt{2}-1) T _0+1 \times 2 R \times(\sqrt{2}-1) T _0$
$\frac{ Q }{ R }=2(3 \sqrt{2}-1)+2(\sqrt{2}-1)=8 \sqrt{2}-4$
Download our appand get started for free
Similar Questions
- 1$P_i, V_i$ and $P_f$, $V_f$ are initial and final pressures and volumes of a gas in a thermodynamic process respectively. If $PV^n =$ constant, then the amount of work done isView Solution
- 2A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$View Solution
- 3An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
- 4The $P-V$ diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process $CD$ is (use $\gamma=1.4$) (in $J$)View Solution
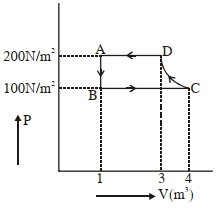
- 5View SolutionIn the above thermodynamic process, the correct statement is
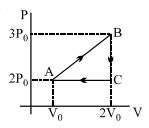
- 6An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
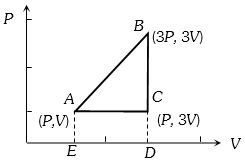
- 7One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)View Solution
- 8Work done for the process shown in the figure is ............ $J$View Solution
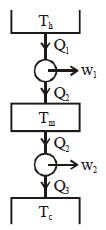
- 9Suppose that two heat engines are connected in series, such that the heat exhaust of the first engine is used as the heat input of the second engine as shown in figure. The efficiencies of the engines are $\eta_1$ and $\eta_2$, respectively. The net efficiency of the combination is given byView Solution
- 10One mole of a perfect gas in a cylinder fitted with a piston has a pressure $P,$ volume $V$ and temperature $T.$ If the temperature is increased by $1 \,K$ keeping pressure constant, the increase in volume isView Solution
