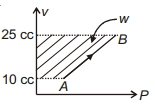
Work done for the process shown in the figure is ............ $J$

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas at an initial temperature of $T\, K$ does $6\, R\, joules$ of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $\frac{5}{3}$ , the final temperature of gas will beView Solution
- 2View SolutionIrreversible process is
- 3Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
- 4Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 5An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution
- 6View SolutionIn an isothermal expansion
- 7If heat given to a system is $6 \,kcal$ and work done is $ 6 kJ.$ Then change in internal energy is ....... $kJ$View Solution
- 8Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 9A Carnot engine with sink's temperature at $17\,^oC$ has $50\%$ efficiency. By how much should its source temperature be changed to increases its efficiency to $60\%$ ?...... $K$View Solution
- 10The adiabatic elasticity of hydrogen gas $(\gamma = 1.4)$ at $NTP$ isView Solution