Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Find the change in the entropy in the following process $100 \,gm$ of ice at $0°C$ melts when dropped in a bucket of water at $50°C$ (Assume temperature of water does not change) ..... $ cal/K$View Solution
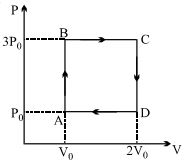
- 2A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution
- 3Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 4A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
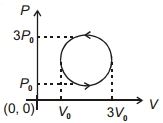
- 5View SolutionWork done in the cyclic process shown in figure is ...........
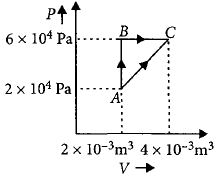
- 6Figure below shows two paths that may be taken by a gas to go from a state $A$ to a state $C.$ In process $AB,$ $400 \,J$ of heat is added to the system and in process $BC,$ $100\, J$ of heat is added to the system. The heat absorbed by the system in the process $AC$ will be ...... $J$View Solution
- 7View SolutionA measure of the degree of disorder of a system is known as
- 8View SolutionEntropy of a thermodynamic system does not change when this system is used for
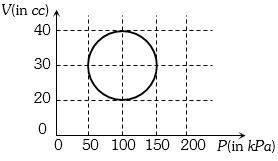
- 9View SolutionA system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is
- 10View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
