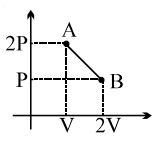
$\frac{P-2 p}{V-v}=\frac{2 p-p}{v-2 v}$
$P-2 p=V \times\left(\frac{-p}{v}\right)$
$\therefore$ equation of the line : $P=V \times\left(\frac{-p}{v}\right)+2 p$
We know that internal Energy $(U)=P V$
$U=P \times V=V \times\left(V \times\left(\frac{-p}{v}\right)+2 p\right)$
$U=-V^{2} \times\left(\frac{p}{v}\right)+2 p V$
As we go from point $A$ to point $B$ volume increases.
As volume increases internal energy initially increases and then decreases, since the internal energy as a function of volume is a downward parabola.
Download our appand get started for free
Similar Questions
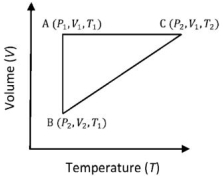
- 1A reversible cyclic process for an ideal gas is shown below. Here, $P , V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$ $(B)$ $W _{ BC }= P _2\left( V _2- V _1\right)$ and $q _{ BC }= H _{ AC }$ $(C)$ $\Delta H _{ CA }<\Delta U _{ CA }$ and $q _{ AC }=\Delta U _{ BC }$ $(D)$ $q_{B C}=\Delta H_{A C}$ and $\Delta H_{C A}>\Delta U_{C A}$
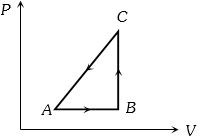
- 2The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
- 3Given below are two statements:View Solution
Statement $I:$ If heat is added to a system, its temperature must increase.
Statement $II:$ If positive work is done by a system in a thermodynamic process, its volume must increase.
In the light of the above statements, choose the correct answer from the options given below
- 4Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
- 5The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 6When an ideal gas in a cylinder was compressed isothermally by a piston, the work done on the gas was found to be $1.5 \times {10^4}\;joules$. During this process aboutView Solution
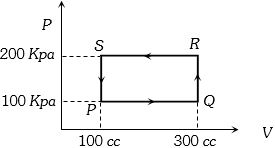
- 7A thermodynamic system is taken through the cycle $PQRSP$ process. The net work done by the system is ....... $J$View Solution
- 8$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
- 9In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
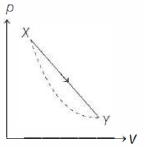
- 10In the $p-V$ diagram below, the dashed curved line is an adiabat.For a process that is described by a straight line joining two points $X$ and $Y$ on the adiabat (solid line in the diagram) heat is (Hint consider the variation in temperature from $X$ to $Y$ along the straight line)View Solution