If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$
Easy
(b)
The efficiency of the cycle is given as $\eta=1-\frac{T_2}{T_1}$. Here $T_1$ is the temperature of the source and $T_2$ is the temperature of the sink. If $T_2=0$ we get efficiency as $\eta$ as $100 \%$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 J$ of heat and $8J$ of work is done on the gas. If the initial internal energy of the gas was $30J.$ The final internal energy will be ...... $J$View Solution
- 2In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gasView Solution
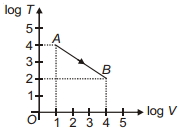
- 3Figure shows, the adiabatic curve on a $\log T$ and log $V$ scale performed on ideal gas. The gas is ............View Solution
- 4View SolutionIn adiabatic expansion of a gas
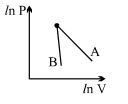
- 5The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
- 6View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
- 7$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
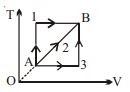
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 8A given mass of a gas expands from a state $A$ to the state $B$ by three paths $1, 2$ and $3$ as shown in $T-V$ indicator diagram. If $W_1, W_2$ and $W_3$ respectively be the work done by the gas along the three paths, thenView Solution
- 9$PV$ curve for the process whose $VT$ curve isView Solution
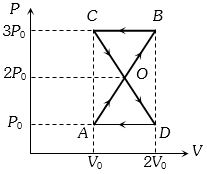
- 10A thermodynamic system undergoes cyclic process $ABCDA $ as shown in figure. The work done by the system in the cycle isView Solution