A diatomic ideal gas is used in a carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\,V$ , the efficiency of the engine is
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
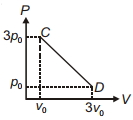
- 1The process $C D$ is shown in the diagram. As system is taken from $C$ to $D$, what happens to the temperature of the system?View Solution
- 2One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$View Solution
- 3Two moles of an ideal monoatomic gas occupies a volume $V$ at $27^o C$. The gas expands adiabatically to a volume $2\ V$. Calculate $(a)$ the final temperature of the gas and $(b)$ change in its internal energy.View Solution
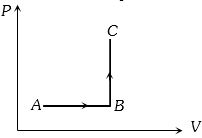
- 4The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done by the system in going from $A \to B \to C$ is $30J$ and $40J$ heat is given to the system. The change in internal energy between $A$ and $C$ is ....... $J$View Solution
- 5An ideal gas in a cylinder is separated by a piston in such a way that the entropy of one part is $S_{1}$ and that of the other part is $S_{2}$. Given that $S _{1}> S _{2}$. If the piston is removed then the total entropy of the system will be :View Solution
- 6Consider two containers $A$ and $B$ containing identical gases at the same pressure, volume and temperature. The gas in container $A$ is compressed to half of its original volume isothermally while the gas in container $B$ is compressed to half of its original value adiabatically. The ratio of final pressure of gas in $B$ to that of gas in $A$ isView Solution
- 7A thermally insulated rigid container contains an ideal gas heated by a filament of resistance $100 \,\Omega$ through a current of $1\,A$ for $5$ min then change in internal energy is...... $kJ$View Solution
- 8View SolutionThe isothermal bulk modulus of a perfect gas at normal pressure is
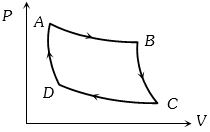
- 9The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
- 10A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
