Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is $3$ times the amount of work done by the cycle, the efficiency of the cycle isView Solution
- 2A gas at initial temperature $T$ undergoes sudden expansion from volume $V$ to $2 \,V$. Then,View Solution
- 3The volume of an ideal gas is $1$ litre and its pressure is equal to $72cm$ of mercury column. The volume of gas is made $900\, cm^3$ by compressing it isothermally. The stress of the gas will be ...... $cm$ (mercury)View Solution
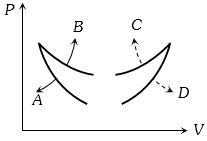
- 4Four curves $A, B, C$ and $D$ are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes areView Solution
- 5$540$ calories of heat convert $1 $ cubic centimeter of water at ${100^o}C$ into $1671 $ cubic centimeter of steam at ${100^o}C$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly ...... $cal$View Solution
- 6View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
- 7For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
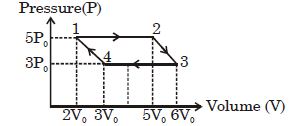
- 8An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
- 9During the melting of a slab of ice at $273\, K$ at atmospheric pressureView Solution
- 10The initial pressure and volume of an ideal gas are $P_0$ and $V_0$. The final pressure of the gas when the gas is suddenly compressed to volume $\frac{ V _0}{4}$ will be (Given $\gamma=$ ratio of specific heats at constant pressure and at constant volume)View Solution
