The specific heat of a gas at constant volume is $21.2\, J/mole/°C.$ If the temperature is increased by $1°C$ keeping the volume constant, the change in its internal energy will be ...... $J$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1This question has Statement $- 1$ and Statement $-2$. Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1$ : The internal energy of a perfect gas is entirely kinetic and depends only on absolute temperature of the gas and not on its pressure or volume.
Statement $2$ : A perfect gas is heated keeping pressure constant and later at constant volume. For the same amount of heat the temperature of the gas at constant pressure is lower than that at constant volume. - 2Aclosed vessel contains a mixture of two diatomic gases Aand $B$. Molar mass of Ais $16$ times that of $B$ and mass of gas $A$ contained in the vessel is $2$ times that of $B$. The following statements are givenView Solution
$(i)$Average kinetic energy per molecule of $A$ is equal to that of $B$.
$(ii)$ Root mean square value of translational velocity of $B$ is four times that of $A$.
$(iii)$ Pressure exerted by $B$ is eight times of that exerted by $A$.
$(iv)$ Number of molecules of $B$ in the cylinder is eight time that of $A$.
- 3One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
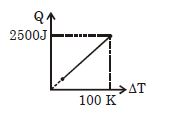
- 4A gaseous mixture consists of $16\,g$ of helium and $16\,g$ of oxygen. The ratio $\frac{{{C_P}}}{{{C_V}}}$ of the mixture isView Solution
- 5A gas is collected over the water at $25°C.$ The total pressure of moist gas was $735 \,mm$ of mercury. If the aqueous vapour pressure at $25°C$ is $23.8\, mm.$ Then the pressure of dry gas is..... $mm$View Solution
- 6The mean free path of molecules of a certain gas at $STP$ is $1500\,d$, where $d$ is the diameter of the gas molecules. While maintaining the standard pressure, the mean free path of the molecules at $373\,K$ is approximately $..........\,d$View Solution
- 7View SolutionThe specific heat of a gas
- 8View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
- 9The density of a gas at normal pressure and $27°C$ temperature is $24.$ Keeping the pressure constant, the density at $127°C$ will beView Solution
- 10The molar specific heat at constant pressure of an ideal gas is $(7/2)R.$ The ratio of specific heat at constant pressure to that at constant volume isView Solution
