Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a carnot engine, the temperature of reservoir is $527^{\circ} C$ and that of $\operatorname{sink}$ is $200 \; K$. If the workdone by the engine when it transfers heat from reservoir to sink is $12000 \; kJ$, the quantity of heat absorbed by the engine from reservoir is $\times 10^{6} \; J$View Solution
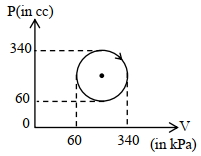
- 2View SolutionThe heat absorbed by a system in going through the given cyclic process is:
- 3View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
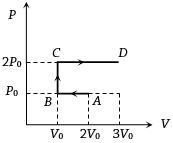
- 4$P-V$ diagram of an ideal gas is as shown in figure. Work done by the gas in process $ABCD$ isView Solution
- 5$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 6Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 7A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
- 8View SolutionWhich of the following is a slow process
- 9View SolutionAn ideal gas, undergoing adiabatic change, has which of the following pressure temperature relationship?
- 10A Carnot engine operates between ${227^o}C$ and ${27^o}C.$ Efficiency of the engine will beView Solution
