Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
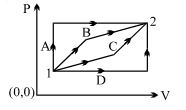
- 1An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
- 2A heat engine operates between a cold reservoir at temperature ${T}_{2}=400\, {K}$ and a hot reservoir at temperature ${T}_{1} .$ It takes $300 \,{J}$ of heat from the hot reservoir and delivers $240\, {J}$ of heat to the cold reservoir in a cycle. The minimum temperature of the hot reservoir has to be $....{K}$View Solution
- 3The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
- 4View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally, if the two gases are compressed to the same final volume
- 5A Carnot engine operating between two reservoirs has efficiency $\frac{1}{3}$. When the temperature of cold reservoir raised by $x$, its efficiency decreases to $\frac{1}{6}$. The value of $x$, if the temperature of hot reservoir is $99^{\circ}\,C$, will be $........\,K$View Solution
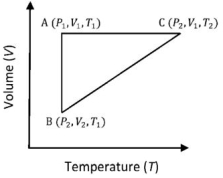
- 6A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
- 7$1\,kg$ of water at $100\, ^{\circ}C$ is converted into steam at $100^{\circ}\,C$ by boiling at atmospheric pressure. The volume of water changes from $1.00 \times 10^{-3}\,m ^3$ as a liquid to $1.671\,m ^3$ as steam. The change in internal energy of the system during the process will be $........kJ$ (Given latent heat of vaporisaiton $=2257\,kJ / kg$. Atmospheric pressure $=1 \times 10^5\,Pa$ )View Solution
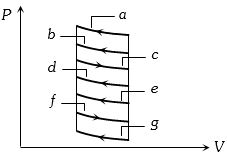
- 8The $P-V$ diagram shows seven curved paths (connected by vertical paths) that can be followed by a gas. Which two of them should be parts of a closed cycle if the net work done by the gas is to be at its maximum valueView Solution
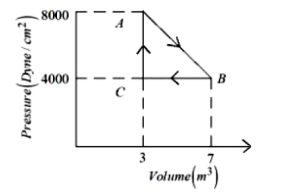
- 9A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :View Solution
- 10View SolutionWhich of the following is incorrect regarding the first law of thermodynamics
