$Assertion :$ The isothermal curves intersect each other at a certain point.
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope.
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope.
AIIMS 2001,AIIMS 2008, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The pressure and density of a diatomic gas $(\gamma = 7/5)$ change adiabatically from $(P, d)$ to $(P', d')$. If $\frac{{d'}}{d} = 32$, then $\frac{{P'}}{P}$ should beView Solution
- 2An ideal Carnot engine, whose efficiency is $40 \%$ receives heat at $500\; K$. If its efficiency is $50 \%$ then the intake temperature for the same exhaust temperature is ......... $K$View Solution
- 3A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
- 4Consider the efficiency of Carnot's engine is given by $\eta=\frac{\alpha \beta}{\sin \theta} \log _{e} \frac{\beta x}{k T}$, where $\alpha$ and $\beta$ are constants. If $T$ is temperature, $k$ is Boltzman constant, $\theta$ is angular displacement and $x$ has the dimensions of length. Then, choose the incorrect option.View Solution
- 5An ideal gas of mass $m$ in a state $A$ goes to another state $B$ via three different processes as shown in figure. If $Q_{1}, Q_{2}$ and $Q_{3}$ denote the heat absorbed by the gas along the three paths, thenView Solution
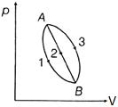
- 6View SolutionWhich is incorrect
- 7View SolutionTwo gases are said to be in thermal equilibrium when they have same
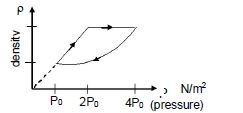
- 8An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 9View SolutionThe internal energy of an ideal gas depends upon
- 10Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$
