$P-V$ diagram of an ideal gas is as shown in figure. Work done by the gas in process $ABCD$ is
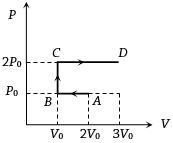

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution
- 2View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
- 3An ideal gas, initially in state $\left( P _{12}, V _1, T _1\right)$ is expanded isobarically to $\left( P _{12}, V _2, T _2\right)$, then adiabatically $\left( P _{34}, V _3, T _3\right)$. It is then contracted isobarically to $\left( P _{34}, V _4, T _4\right)$ and finally adiabatically back to the initial state. The efficiency of this cycle isView Solution
- 4A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............View Solution
- 5A diatomic gas with rigid molecules does $10\, J$ of work when expanded at constant pressure. What would be the heat energy absorbed by the gas, in this process ..... $J$.View Solution
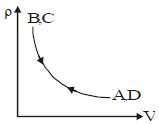
- 6Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution
- 7A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 8View SolutionFirst law of thermodynamics is a special case of
- 9A solid body of constant heat capacity $1\ J/^o C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with $8$ reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^o C$ to final temperature $200^o C$. Entropy change of the body in the two cases respectively is :
- 10How much work to be done in decreasing the volume of and ideal gas by an amount of $2.4 \times {10^{ - 4}}{m^3}$ at normal temperature and constant normal pressure of .......$joule$ $1 \times {10^5}N/{m^2}$View Solution
