Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ is


AIEEE 2009, Medium
Work done by the system in the isothermal process
$DA\,is\,W = 2.303nRT\,{\log _{10}}\frac{{{P_D}}}{{{P_A}}}$
$ = 2.303 \times 2\,R \times 300\,{\log _{10}}\frac{{1 \times {{10}^5}}}{{2 \times {{10}^5}}} = - 414R$
Therefore work done on the gas is $+414\,R.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
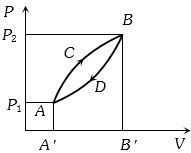
- 1A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the areaView Solution
- 2View SolutionIn a cyclic process, the internal energy of the gas
- 3View SolutionAn ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
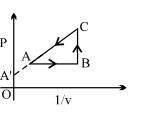
- 4View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
- 5In an isochoric process if ${T_1} = {27^o}C$ and ${T_2} = {127^o}C,$ then ${P_1}/{P_2}$ will be equal toView Solution
- 6View SolutionFirst law thermodynamics states that
- 7$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution
- 8View SolutionIf an ideal gas is compressed isothermally then
- 9A sample of $0.1\, g$ of water at $100^o C$ and normal pressure $(1.013 \times 10^5 N m^{-2} )$ requires $54\ cal $ of heat energy to convert to steam at $100^o C.$ If the volume of the steam produced is $167.1 \,cc,$ the change in internal energy of the sample, is ....... $J$View Solution
- 10Two cylinders contain same amount of ideal monatomic gas. Same amount of heat is given to two cylinders. If temperature rise in cylinder $A$ is $T_0$ then temperature rise in cylinder $B$ will be .........View Solution