For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):
JEE MAIN 2021, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
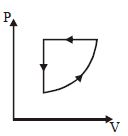
- 1For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
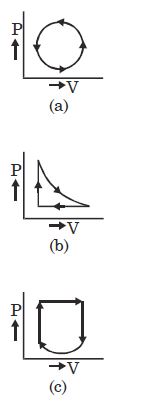
- 2View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure
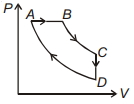
- 3For $P-V$ diagram of a thermodynamic cycle as shown in figure, process $B C$ and $D A$ are isothermal. Which of the corresponding graphs is correct?View Solution
- 4A litre of dry air at $STP$ expands adiabatically to a volume of $3$ litres. If $\gamma=1.40,$ the work done by air is$(3^{1.4}=4.6555)$ [Take air to be an ideal gas $]$View Solution
- 5The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 6A thermodynamic process is the pressure and volumes corresponding to some points in the figure are, $P_A = 3 \times 10^4 Pa$, $V_A = 2 \times 10^{-3}\, m^3$, $P_B = 8 \times 10^4 Pa$, $V_D = 5 \times 10^{-3}\,m^3$. In process $AB, 600\, J$ of heat and in process $BC, 200\, J$ of heat is added to the system. The change in the internal energy in process $AC$ would be .... $J$View Solution
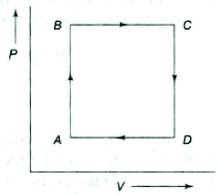
- 7View SolutionDuring the thermodynamic process shown in figure for an ideal gas
- 8The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal toView Solution
- 9$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
- 10For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will beView Solution
