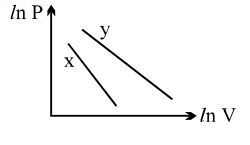
For two different gases $X$ and $Y$, having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{V1}$ and $C_{V2}$ respectively, the ln $P$ versus ln $V$ graph is plotted for adiabatic process, as shown

Advanced
since the two equations are of the form, $\ln P+m \ln V=C$
Hence, $P V^{m}=$ constant
Thus, slope of the graph is equal to $\gamma$
$\gamma=\frac{f+2}{f}=1+\frac{2}{f}$
since $\gamma_{1}>\gamma_{2},$ thus $f_{2}>f_{1}$
since $C_{V}=\frac{f}{2} R,$ thus $C_{V_{2}}>C_{V_{1}}$
Answer is $\mathrm{B}, \mathrm{C}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
- 2Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$View Solution
- 3View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
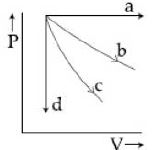
- 4The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process$(es)$ does the temperature of the gas decrease?View Solution
- 5Column $I$ Contains a list of processes involving expansion of an ideal gas. Match this with Column $II$ describing the thermodynamic change during this process. Indicate your answer by darkening the appropriate bubbles of the $4 \times 4$ matrix given in the $ORS$.View Solution
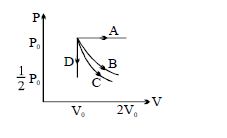
Column $I$ Column $II$ $(A)$ An insulated container has two chambers separated by a valve. Chamber $I$ contains an ideal gas and the Chamber $II$ has vacuum. The valve is opened. $(p)$ The temperature of the gas decreases $(B)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^2}$, where $\mathrm{V}$ is the volume of the gas $(q)$ The temperature of the gas increases or remains constant $(C)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^{4 / 3}}$, where $\mathrm{V}$ is its volume $(r)$ The gas loses heat $(D)$ An ideal monoatomic gas expands such that its pressure $\mathrm{P}$ and volume $\mathrm{V}$ follows the behaviour shown in the graph $Image$ $(s)$ The gas gains heat 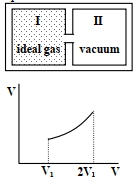
- 6One mole of an ideal gas expands adiabatically from an initial state $\left(T_A, V_0\right)$ to final state $\left(T_f, 5 V_0\right)$. Another mole of the same gas expands isothermally from a different initial state ( $T_{\mathrm{B}}, \mathrm{V}_0$ ) to the same final state $\left(T_{\mathrm{f}}, 5 V_0\right)$. The ratio of the specific heats at constant pressure and constant volume of this ideal gas is $\gamma$. What is the ratio $T_{\mathrm{A}} / T_{\mathrm{B}}$ ?View Solution
- 7During an adiabatic compression, $830\, J$ of work is done on $2\, moles$ of a diatomic ideal gas to reduce its volume by $50\%$. The change in its temperahture is nearly..... $K$ $(R\, = 8.3\, J\,K^{-1}\, mol^{-1} )$View Solution
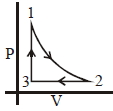
- 8Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
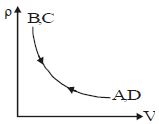
- 9Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution
- 10Two identical balls, $A$ and $B$ , of uniform composition and initially at the same temperature, each absorb exactly the same amount of heat. $A$ is hanging down from the ceiling while $B$ rests on the horizontal floor in the same room. Assuming no subsequent heat loss by the balls, which of the following statements is correct about their final temperatures, $T_A$ and $T_B$ , once the balls have reached their final state?View Solution