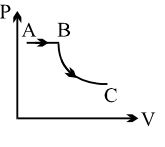
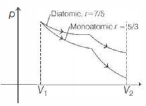
One mole of a monoatomic gas and one mole of a diatomic gas are initially in the same state. Both gases are expanded isothermally and then adiabatically, such that they acquire the same final state. Choose the correct statement.
KVPY 2018, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
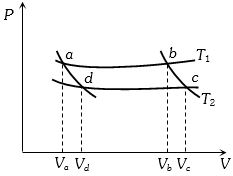
- 1In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
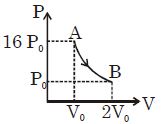
- 2Figure shows a polytropic process for an ideal gas. The work done by the gas will be in process $AB$ isView Solution
- 3View SolutionThe work done in which of the following processes is zero
- 4View SolutionIn thermodynamic processes which of the following statements is not true?
- 5View SolutionThe internal energy of an ideal gas depends upon
- 6View SolutionIf an ideal gas is compressed isothermally then
- 7The temperature of a hypothetical gas increases to $\sqrt 2 $ times when compressed adiabatically to half the volume. Its equation can be written asView Solution
- 8View SolutionWhen an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is
- 9One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ isView Solution
- 10View SolutionA process is shown in the diagram. Which of the following curves may represent the same process ?