Download our appand get started for free
Similar Questions
- 1Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason $R$.View Solution
Assertion $A$ : If $dQ$ and $dW$ represent the heat supplied to the system and the work done on the system respectively. Then according to the first law of thermodynamics $d Q=d U-d W$.
Reason $R :$ First law of thermodynamics is based on law of conservation of energy.
In the light of the above statements, choose the correct answer from the option given below :
- 2An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 3An iron rod of heat capacity $C$ is heated to temperature $8T_0$ . It is then put in a cylindrical vessel of adiabatic walls having two moles of air which can be treated as diatomic ideal gas at temperature $T_0$ and closed by a movable piston which is also adiabatic. The atmospheric pressure is $P_0$ . The cylinder with the piston combined have heat capacity $2C$ . Find the equilibrium temperature . (Assume temperature of air to be uniform and equal to vessel at all times) .View Solution
- 4Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
- 5An ideal gas expands isothermally along $ab$ and does $600\,J$ of work. During the processView Solution
- 6Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
- 7During the adiabatic expansion of $2 \,moles$ of a gas, the internal energy was found to have decreased by $100 J$. The work done by the gas in this process is ..... $J$View Solution
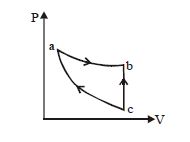
- 8View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
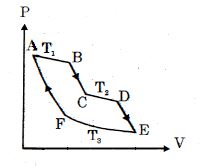
- 9One mole of ideal gas taken through a cycle process with alternate isothermal and adiabatic curves. In $P-V$ diagram $AB, CD, EF$ are isothermal curves at the absolute temperature $T_1, T_2$ and $T_3$ respectively and $BC, DE$ and $FA$ are adiabatic curves respectively. If $\frac{{{V_B}}}{{{V_A}}} = 2,\,\frac{{{V_D}}}{{{V_C}}} = 2$ then for cycle is shown in figure four statements are being made given below. (Figure is not drawn on scale)View Solution
Statement $1$ : Ratio of volumes $\frac{{{V_E}}}{{{V_F}}} = 4$
Statement $2$ : Magnitude of work done in isothermal compression $EF$ is $2RT_3\ ln\ (2)$
Statement $3$ : Ratio of heat supplied to gas in the process $AB$ to heat rejected by gas in process $EF$ is $\frac{{{T_1}}}{{2{T_3}}}$
Statement $4$ : Net work done by gas in the cycle $ABCDEFA$ is $(T_1 + T_2 - 2T_3) R\ ln\ (2)$
Find the number of correct statement $(s)$ given for the cyclic process followed by gas
- 10$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes.
