$Assertion :$ In free expansion of an ideal gas, the entropy increases.
$Reason :$ Entropy increases in all natural processes.
$Reason :$ Entropy increases in all natural processes.
AIIMS 2007, Easy
In free expansion of an ideal gas, work done comes from internal energy of the gas and since randomness increases or Gibbs free energy increases so we can say entropy increases.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
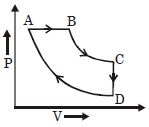
- 2A cyclic process $ABCD$ is shown in the given $P-V$ diagram. $P-T$ diagram that represents the same process isView Solution
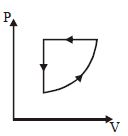
- 3For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
- 4View SolutionWork done on or by a gas, in general depends upon the
- 5A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
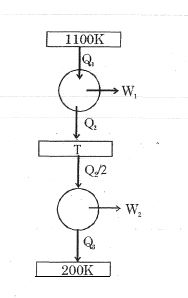
- 6Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
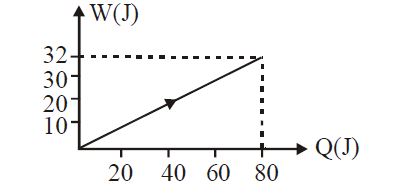
- 7View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
- 8View SolutionCan two isothermal curves cut each other
- 9An engine has an efficiency of $1/6.$ When the temperature of sink is reduced by $62^o C,$ its efficiency is doubled. Temperatures of the source is ....... $^oC$View Solution
- 10If minimum possible work is done by a refrigerator in converting $100\; grams$ of water at $0^{\circ} C$ to ice, how much heat (in calories) is released to the surrounding at temperature $27^{\circ} C$ (Latent heat of ice $=80 Cal / gram$ ) to the nearest integer?View Solution
